技术资料
-
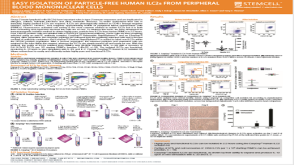 科学海报Easy Isolation of Particle-Free Human ILC2s from Peripheral Blood Mononuclear Cells
科学海报Easy Isolation of Particle-Free Human ILC2s from Peripheral Blood Mononuclear CellsConference:
AAI 2018
发布日期: 11/12/2019 -
 科学海报Efficient Enrichment of Functional ILC Subsets from Human PBMCs by Immunomagnetic Selection
科学海报Efficient Enrichment of Functional ILC Subsets from Human PBMCs by Immunomagnetic SelectionConference:
AAI 2018
发布日期: 10/25/2019


 EasySep™小鼠TIL(CD45)正选试剂盒
EasySep™小鼠TIL(CD45)正选试剂盒







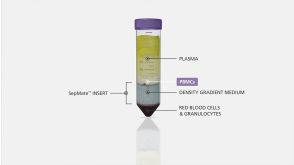
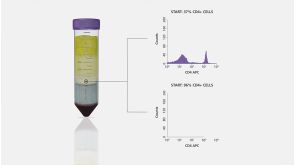
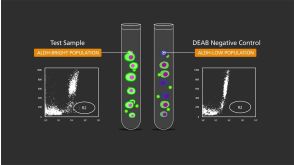


 沪公网安备31010102008431号
沪公网安备31010102008431号