技术资料
-
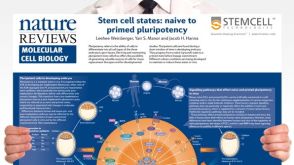 挂图Stem Cell States: Naive to Primed Pluripotency Properties of naive (ground) and primed pluripotent stem cells
挂图Stem Cell States: Naive to Primed Pluripotency Properties of naive (ground) and primed pluripotent stem cells -
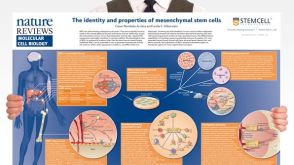 挂图The Identity and Properties of Mesenchymal Stem Cells Overview of MSC expansion, differentiation, immunoregulatory properties and therapeutic potential
挂图The Identity and Properties of Mesenchymal Stem Cells Overview of MSC expansion, differentiation, immunoregulatory properties and therapeutic potential -
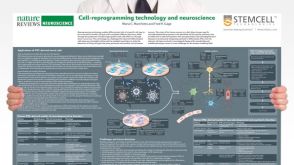 挂图Cell-Reprogramming Technology and Neuroscience Details on human iPSC-derived models of neuropsychiatric and neurodegenerative disorders
挂图Cell-Reprogramming Technology and Neuroscience Details on human iPSC-derived models of neuropsychiatric and neurodegenerative disorders -
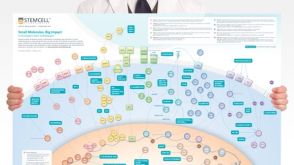 挂图Small Molecules, Big Impact in Pluripotent Stem Cell Research Overview of signaling pathways and small molecules in pluripotent stem cell research
挂图Small Molecules, Big Impact in Pluripotent Stem Cell Research Overview of signaling pathways and small molecules in pluripotent stem cell research -
 挂图Identification of Colonies Derived from Mouse Hematopoietic Progenitors Representative colony images and tips for identifying progenitor subtypes in CFU assays
挂图Identification of Colonies Derived from Mouse Hematopoietic Progenitors Representative colony images and tips for identifying progenitor subtypes in CFU assays -
 挂图Pluripotent Stem Cell Biology Nuclear transfer, reprogramming and cell fusion to induce pluripotency in somatic cells
挂图Pluripotent Stem Cell Biology Nuclear transfer, reprogramming and cell fusion to induce pluripotency in somatic cells -
 挂图Neural Stem Cells Overview of the types of NSCs and their potential use as therapeutic agents for disease
挂图Neural Stem Cells Overview of the types of NSCs and their potential use as therapeutic agents for disease -
 挂图Directed Differentiation of Pluripotent Stem Cells Strategies for differentiating ES and iPS cells into ectoderm, endoderm or mesoderm
挂图Directed Differentiation of Pluripotent Stem Cells Strategies for differentiating ES and iPS cells into ectoderm, endoderm or mesoderm


 EasySep™小鼠TIL(CD45)正选试剂盒
EasySep™小鼠TIL(CD45)正选试剂盒








 沪公网安备31010102008431号
沪公网安备31010102008431号