搜索结果: 'methocult media formulations for human hematopoietic cells serum containing'
-
 科学海报An Animal Component-Free, Serum-Free Culture Method for Generation of Human Dendritic Cells
科学海报An Animal Component-Free, Serum-Free Culture Method for Generation of Human Dendritic Cells产品类型:
Conference:
AAI 2016
产品号#:
10985
10986
10987
10988
10989
产品名:
ImmunoCult™ 树突状细胞培养试剂盒
ImmunoCult™-ACF树突状细胞培养基
ImmunoCult™-ACF树突状细胞培养基
ImmunoCult™-ACF树突状细胞分化添加物
ImmunoCult™树突状细胞成熟添加物
-
 技术公告Flow Cytometry Methods for Identifying Mouse Hematopoietic Stem and Progenitor Cells
技术公告Flow Cytometry Methods for Identifying Mouse Hematopoietic Stem and Progenitor Cells产品类型:
细胞类型:
造血干/祖细胞
产品号#:
产品名:
-
 50:59
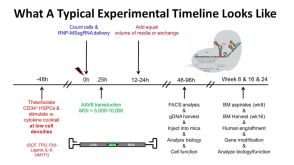
线上讲座Gene Targeting in Hematopoietic Stem Cells for Basic and Translational Research发布日期: 11/28/2018
50:59
线上讲座Gene Targeting in Hematopoietic Stem Cells for Basic and Translational Research发布日期: 11/28/2018


 EasySep™小鼠TIL(CD45)正选试剂盒
EasySep™小鼠TIL(CD45)正选试剂盒








 沪公网安备31010102008431号
沪公网安备31010102008431号