Induced pluripotent stem (iPS) cells are a valuable resource for discovery of epigenetic changes critical to cell type-specific differentiation. Although iPS cells have been generated from other terminally differentiated cells,the reprogramming of normal adult human basal prostatic epithelial (E-PZ) cells to a pluripotent state has not been reported. Here,we attempted to reprogram E-PZ cells by forced expression of Oct4,Sox2,c-Myc,and Klf4 using lentiviral vectors and obtained embryonic stem cell (ESC)-like colonies at a frequency of 0.01%. These E-PZ-iPS-like cells with normal karyotype gained expression of pluripotent genes typical of iPS cells (Tra-1-81,SSEA-3,Nanog,Sox2,and Oct4) and lost gene expression characteristic of basal prostatic epithelial cells (CK5,CK14,and p63). E-PZ-iPS-like cells demonstrated pluripotency by differentiating into ectodermal,mesodermal,and endodermal cells in vitro,although lack of teratoma formation in vivo and incomplete demethylation of pluripotency genes suggested only partial reprogramming. Importantly,E-PZ-iPS-like cells re-expressed basal epithelial cell markers (CD44,p63,MAO-A) in response to prostate-specific medium in spheroid culture. Androgen induced expression of androgen receptor (AR),and co-culture with rat urogenital sinus further induced expression of prostate-specific antigen (PSA),a hallmark of secretory cells,suggesting that E-PZ-iPS-like cells have the capacity to differentiate into prostatic basal and secretory epithelial cells. Finally,when injected into mice,E-PZ-iPS-like cells expressed basal epithelial cell markers including CD44 and p63. When co-injected with rat urogenital mesenchyme,E-PZ-iPS-like cells expressed AR and expression of p63 and CD44 was repressed. DNA methylation profiling identified epigenetic changes in key pathways and genes involved in prostatic differentiation as E-PZ-iPS-like cells converted to differentiated AR- and PSA-expressing cells. Our results suggest that iPS-like cells derived from prostatic epithelial cells are pluripotent and capable of prostatic differentiation; therefore,provide a novel model for investigating epigenetic changes involved in prostate cell lineage specification.
View Publication
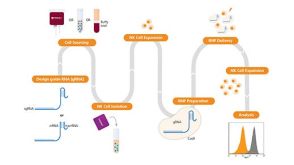
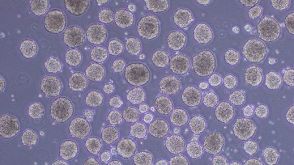 实验方案How to Differentiate hPSCs to Lymphoid-Competent CD34+ Hematopoietic Stem and/or Progenitor Cells Using a Scalable Suspension Protocol
实验方案How to Differentiate hPSCs to Lymphoid-Competent CD34+ Hematopoietic Stem and/or Progenitor Cells Using a Scalable Suspension Protocol

 EasySep™小鼠TIL(CD45)正选试剂盒
EasySep™小鼠TIL(CD45)正选试剂盒








 沪公网安备31010102008431号
沪公网安备31010102008431号