搜索结果: 'methocult media formulations for human hematopoietic cells serum containing'
-
 技术手册Maintenance of Human Pluripotent Stem Cells in mTeSR™ Plus
技术手册Maintenance of Human Pluripotent Stem Cells in mTeSR™ Plus产品类型:
产品号#:
05825
100-0276
产品名:
mTeSR™ Plus
-
 产品手册Derive, Expand, and Differentiate Human Skeletal Muscle Progenitor Cells
产品手册Derive, Expand, and Differentiate Human Skeletal Muscle Progenitor Cells产品类型:
品牌:
MyoCult
产品号#:
产品名:
-
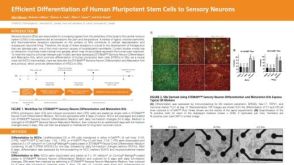 科学海报Efficient Differentiation of Human Pluripotent Stem Cells to Sensory Neurons
科学海报Efficient Differentiation of Human Pluripotent Stem Cells to Sensory Neurons产品类型:
Conference:
ISSCR 2022
产品号#:
产品名:
-
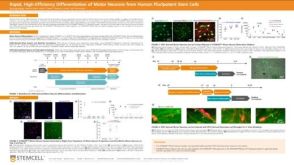 科学海报Rapid, High-Efficiency Differentiation of Motor Neurons from Human Pluripotent Stem Cells
科学海报Rapid, High-Efficiency Differentiation of Motor Neurons from Human Pluripotent Stem Cells产品类型:
Conference:
FENS 2022
产品号#:
产品名:
-
 科学海报Efficient, Reproducible and High-Throughput-Compatible Protocols for Differentiation of Human Pluripotent Stem Cell Lines Into Kidney Organoids
科学海报Efficient, Reproducible and High-Throughput-Compatible Protocols for Differentiation of Human Pluripotent Stem Cell Lines Into Kidney Organoids产品类型:
Conference:
ISSCR 2019
产品号#:
产品名:


 EasySep™小鼠TIL(CD45)正选试剂盒
EasySep™小鼠TIL(CD45)正选试剂盒









 沪公网安备31010102008431号
沪公网安备31010102008431号